Legato Biologics’s upstream process development capabilities are built upon state-of-the-art infrastructure and an experienced team dedicated to delivering high-quality biopharmaceutical products. The pilot facility is equipped with advanced bioreactor systems, including 20L and 50L wave bioreactors and single-use bioreactors ranging from 2L to 500L. These systems are complemented by a broad suite of high-throughput process analytical technologies (PAT) to monitor and control critical process parameters with precision, achieving scalability and consistency from development through clinical manufacturing. This comprehensive platform supports the production of materials for IND registration and clinical studies up to Phase 2.
Adhering to Quality by Design (QbD) principles, Legato Biologics’ systematic upstream process development includes extensive process characterization to identify critical quality attributes (CQAs) and process parameters. Legato delivers robust processes with optimized yields and quality, and thorough technology transfer to ensure seamless scalability across manufacturing sites. By integrating advanced analytics and a risk-based methodology, Legato Biologics guarantees that every stage of process development aligns with regulatory requirements and industry best practices.
The team at Legato Biologics has a proven track record of successfully validating upstream processes for multiple commercialized antibody projects. Their expertise spans from early-stage development to full-scale manufacturing, including process validation for regulatory submissions. By combining innovative technologies with a commitment to excellence, Legato Biologics delivers reliable and scalable upstream processes that meet the stringent demands of the biopharmaceutical industry.
Legato Biologics’ downstream process development platform is equipped with state-of-the-art purification systems designed to achieve high quality and yield. The platform incorporates a comprehensive suite of purification techniques, including affinity chromatography, ion exchange chromatography, hydrophobic chromatography, depth filtration, nanofiltration, and ultrafiltration. These advanced technologies are critical for the robust and scalable purification of complex biomolecules with high purity to meet regulatory and commercial standards.
Legato Biologics’s downstream capabilities have been proven by the successful process development of over 10 molecules, encompassing monoclonal antibodies, bispecific antibodies, and fusion proteins. The platform consistently achieves highly effective purification yields (up to 85%), optimizing product recovery while maintaining critical quality attributes. Through rigorous process optimization and innovation, Legato Biologics tailors its downstream processes to each molecule’s unique characteristics, facilitating robust and reproducible manufacturing.
To mitigate potential supply chain risks and reduce production costs, Legato Biologics has developed extensive expertise in utilizing domestic resins for purification processes. This strategic approach not only enhances supply chain resilience but also provides cost-effective alternatives without compromising performance or regulatory compliance. By leveraging these capabilities, Legato Biologics offers flexible and reliable purification solutions to its partners and clients.
Legato Biologics adheres to the Quality by Design (QbD) principles across all stages of downstream process development, including process characterization, technology transfer, and process validation. This systematic and science-driven approach ensures that every purification step is thoroughly understood and optimized for seamless scale-up and regulatory approval. With a strong commitment to quality and innovation, Legato Biologics continues to deliver robust downstream solutions for commercialized antibody projects, ensuring product consistency and regulatory readiness.
Legato Biologics offers comprehensive analytical capabilities, providing robust support for both clinical and commercial products. The team has extensive experience in conducting release and stability studies that encompass a wide range of testing, including physicochemical, bioactivity, biosafety, and characterization-related assays. These capabilities allow Legato Biologics to meet the stringent regulatory requirements for product filings in China, Europe, and the United States. The company’s expertise extends to specification setting, ensuring that all critical parameters are well-defined to support regulatory approvals and ongoing quality assurance.
A key strength of Legato Biologics’s analytical platform is its standardized sample management workflow. The process includes meticulous sample storage, a well-structured sampling plan, and end-to-end sample and data tracking using electronic management systems. This comprehensive approach enhances traceability, reduces errors, and ensures compliance with regulatory expectations. By implementing electronic data management systems, Legato Biologics maintains data integrity and facilitates efficient collaboration across teams and stakeholders.
Legato Biologics provides a full suite of product characterization capabilities, enabling a deep understanding of the structure, function, heterogeneity, and impurity profiles of biologic products from various modalities. These include monoclonal and multi-specific antibodies, fusion proteins, and antibody-drug conjugates (ADCs). The company’s expertise in characterization ensures that critical quality attributes (CQAs) are thoroughly assessed, supporting both development and regulatory submissions with high-confidence data.
Following the Quality by Design (QbD) framework, Legato Biologics excels in analytical method development, validation, transfer, and lifecycle management. Our experience extends to conducting comparability studies for clinical and post-approval process changes, as well as similarity assessments for biosimilar development. This expertise allows Legato Biologics to support clients in navigating the complex regulatory landscape while high product consistency and quality throughout the product’s lifecycle.

Legato Biologics offers comprehensive analytical and regulatory capabilities, supported by state-of-the-art laboratory equipment and a highly skilled team of professionals. The company’s quality control operations feature advanced instrumentation, including HPLC, UPLC, FQ-PCR spectrometers, capillary electrophoresis apparatus, flow cytometer, and isolator systems. These cutting-edge tools enable precise and reliable testing to support the entire biopharmaceutical development process, from raw material assessment to final product release and stability testing. Legato Biologics’ rigorous monitoring and maintenance programs ensure that all facilities and equipment consistently operate within regulatory standards, maintaining a high level of quality and compliance.
Legato Biologics’s testing capabilities encompass a full range of quality control services, covering raw material analysis, in-process monitoring, final product release, and stability studies. The company’s robust quality systems ensure that all incoming materials, in-process samples, and finished products are tested in accordance with pre-established specifications to secure product consistency and safety. With a strong focus on compliance, Legato Biologics adheres to global regulatory requirements and industry best practices, providing reliable data that supports regulatory submissions and lifecycle management.

On the regulatory front, Legato Biologics offers high-level strategic guidance to help clients navigate the complex regulatory landscape. Their services include regulatory strategy development, gap analysis, and risk assessment to identify potential challenges and ensure a smooth pathway to regulatory approval. The company’s expertise in hands-on dossier preparation includes full drafting of critical sections such as M2 (Clinical Overview), M3 (Quality), and M4 (Nonclinical Study Reports), along with comprehensive data analysis, interpretation, and specification development.
Legato Biologics also provides end-to-end support for Investigational New Drug (IND) submissions, offering in-house electronic Common Technical Document (eCTD) submission capabilities and proactive responses to regulatory inquiries. Our experienced team collaborates closely with clients to ensure timely and successful submissions, addressing regulatory concerns effectively and facilitating project progression. Beyond initial submissions, Legato Biologics supports clients with ongoing lifecycle management, including the preparation and submission of annual reports and amendments to maintain regulatory compliance and address evolving product development needs.
By integrating top-tier analytical capabilities with a strategic regulatory approach, Legato Biologics positions itself as a trusted partner in the biopharmaceutical industry, offering comprehensive solutions to ensure product quality, regulatory success, and long-term market viability.
Legato Biologics offers comprehensive capabilities in Antibody-Drug Conjugate (ADC) development, encompassing the entire spectrum from antibody engineering to GMP manufacturing. With rich experience in antibody development, the company excels in cell line development, process optimization, and excipient and formulation process development. Our expertise extends to analytical method development and validation for robust quality control throughout the ADC development lifecycle. Legato Biologics’ GMP manufacturing capabilities further support seamless transition from early-stage development to clinical and commercial production, adhering to global regulatory standards.
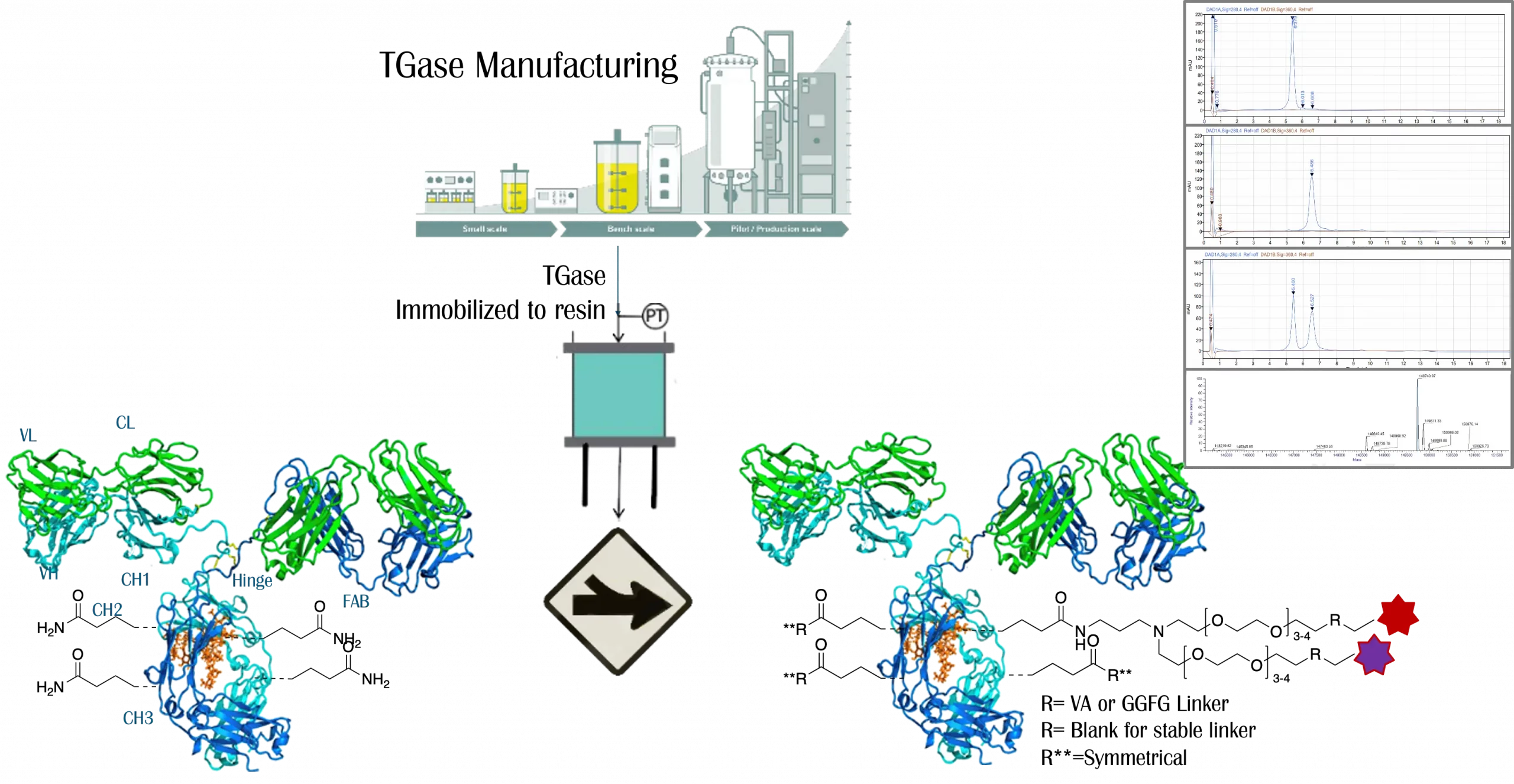
A key strength of Legato Biologics lies in its extensive linker-payload platform, which includes a diverse library of linker-payload combinations. The portfolio features proprietary branched water-soluble linker-payload technology, designed to enhance ADC stability and solubility. Additionally, Legato Biologics has developed proprietary DXd-like linkers and novel DXd-derivatives with improved biological activity and enhanced bystander effects, offering superior therapeutic potential. The company also provides tailored solutions for linker-payload modifications, enabling customized ADC designs to meet specific therapeutic needs.
In conjugation process development, Legato Biologics leverages its proprietary uniQon™ site-specific conjugation platform, which ensures precise and consistent drug-to-antibody ratios (DAR) to optimize therapeutic efficacy and reduce off-target effects. The company’s expertise in excipient and formulation process development allows that ADCs maintain stability and efficacy throughout their shelf life. Additionally, Legato Biologics’ analytical capabilities play a crucial role in method development and validation, supporting regulatory compliance and product characterization.
With a well-established GMP manufacturing infrastructure, Legato Biologics offers end-to-end ADC development support, from early-stage research to large-scale production. Our holistic approach, integrating advanced conjugation technologies, formulation expertise, and rigorous quality assurance, positions Legato Biologics as a strategic partner for companies looking to develop next-generation ADC therapeutics.