Legato Biologics offers end-to-end Chemistry, Manufacturing, and Controls (CMC) services in America, specializing in monoclonal and multi-specific antibody development. With a focus on rapid IND submission, Legato Biologics provides a streamlined pathway from gene synthesis to eCTD filing as swiftly as 11 months, constituting a rapid approach to FDA review. Our expertise in fed-batch processing routinely achieves an industry-leading yield of 4-10 grams per liter, demonstrating our commitment to efficiency and scalability in biologics production.
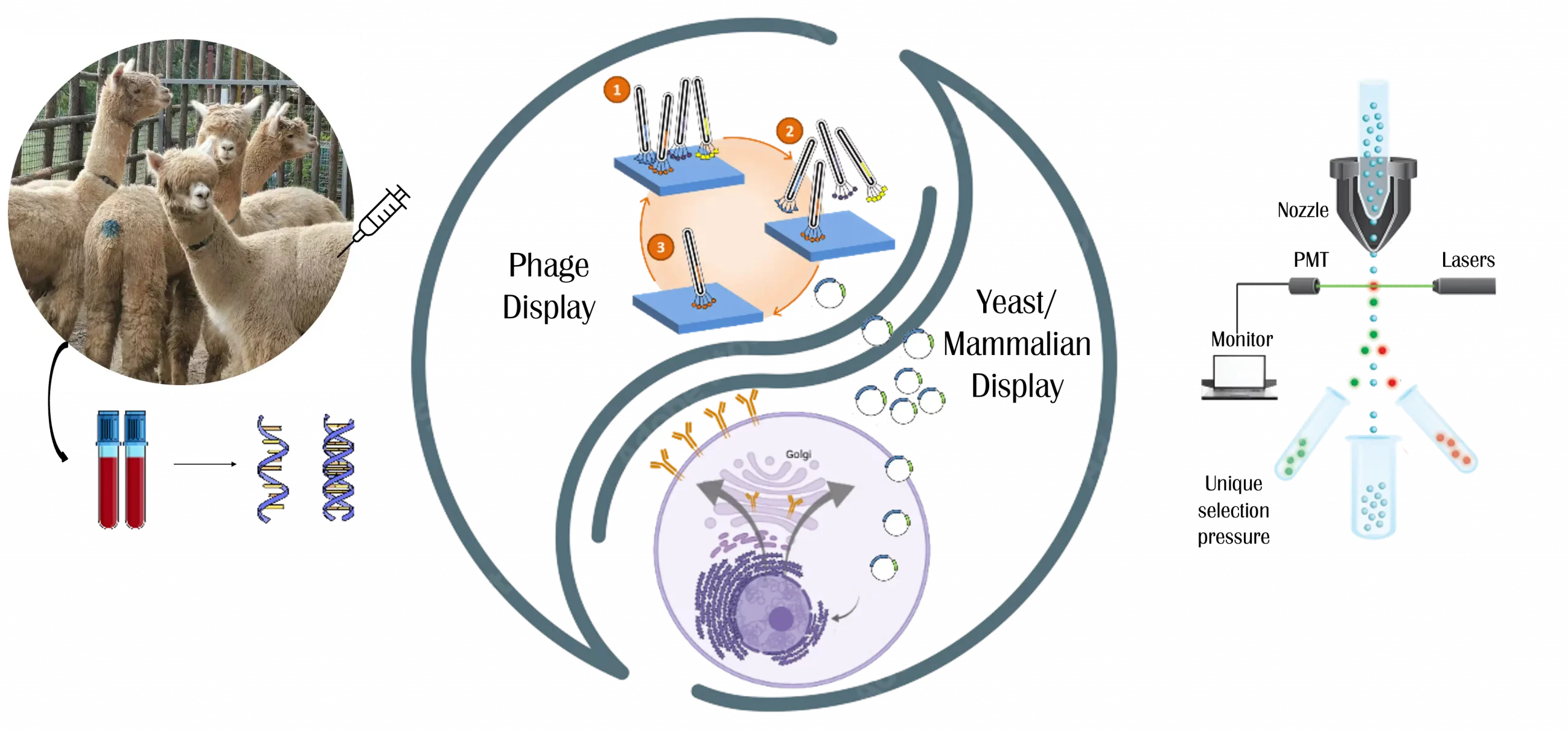
In addition to monoclonal antibodies, Legato Biologics is at the forefront of nanobody discovery and development through its proprietary SparxNano™ platform. As a global leader in the discovery of single-domain antibodies, we offer comprehensive services covering the entire development cycle, from target identification to clinical validation. Our cutting-edge technologies guarantee high-affinity nanobody candidates with superior therapeutic potential across a wide range of disease areas.
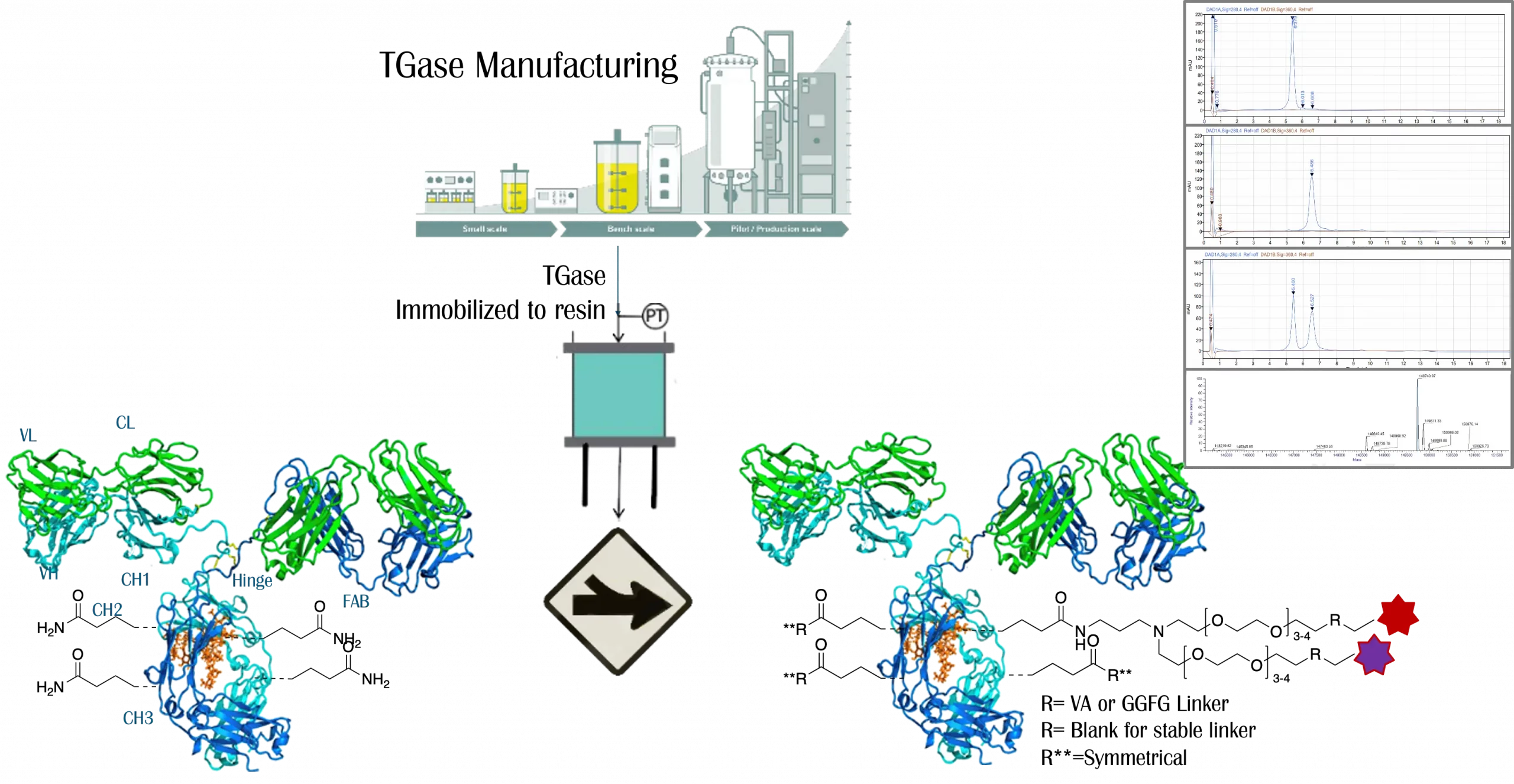
Legato Biologics has a dedicated focus on Antibody-Drug Conjugate (ADC) development, leveraging its proprietary uni–Qon™ site-specific conjugation technology. This innovative platform achieves a drug-to-antibody ratio (DAR) close to 4 with high homogeneity, optimizing therapeutic efficacy while minimizing toxicity. Our comprehensive ADC services cover the entire development lifecycle, from discovery to clinical trials, with capabilities to support both mono- and dual-payload designs, providing customized solutions to meet specific therapeutic needs.
Our CRDMO services provide full support for the entire journey of biologics development, from discovery through Phase 2 clinical trials. Legato Biologics’ integrated platforms allow seamless transitions between key development stages, ensuring alignment with regulatory requirements and project timelines. By combining scientific expertise with state-of-the-art infrastructure, we help clients accelerate their pipeline progress with efficiency and precision.
At Legato Biologics, quality is at the core of our operations. We are dedicated to maintaining the highest standards through meticulous documentation with over 1,000 Standard Operating Procedures (SOPs) guiding every aspect of development and manufacturing. Our independent Quality Assurance (QA) team monitors compliance through rigorous audits and inspections, ensuring adherence to regulatory expectations and industry best practices, giving our clients confidence in the integrity and reliability of our services.